Research
[Clinical Chemistry] Molecular diagnosis of the COVID-19 strain
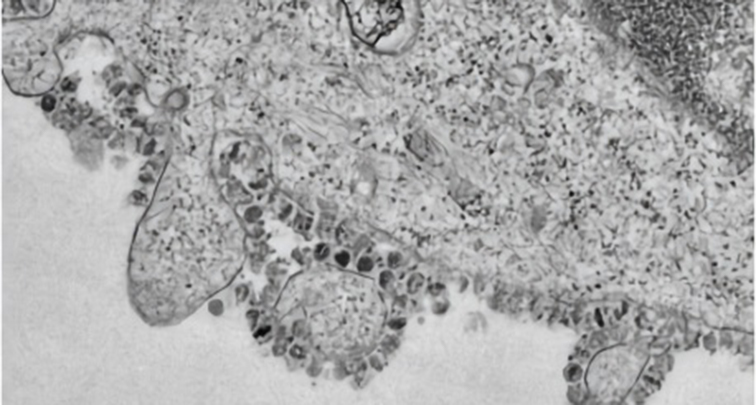
Clinical Chemistry, 31 January 2020
Daniel K W Chu, Yang Pan, Samuel M S Cheng, Kenrie P Y Hui, Pavithra Krishnan, Yingzhi Liu, Daisy Y M Ng, Carrie K C Wan, Peng Yang, Quanyi Wang, Malik Peiris, Leo L M Poon
Highlights
- Outbreaks in health care workers indicate human-to-human transmission. Molecular tests for rapid detection of this virus are urgently needed for the early identification of infected patients.
- Two 1-step quantitative real-time reverse-transcription PCR assays to detect two different regions (ORF1b and N) of the viral genome.
- Using RNA extracted from cells infected by the SARS coronavirus as a positive control, these assays were shown to have a dynamic range of at least seven orders of magnitude (2×10-4 -2000 TCID50/reaction). Using DNA plasmids as positive standards, the detection limits of these assays were found to be below 10 copies per reaction.
- The established assays can achieve rapid detection of COVID-19 in human samples, thereby allowing early identification of patients.
Podcast: Interview with Leo L.M. Poon on Molecular Detection of Novel Human Influenza H1N1

![Photo of [Nature Microbiology] Metallodrug ranitidine bismuth citrate suppresses SARS-CoV-2 replication and relieves virus-associated pneumonia in Syrian hamsters](https://fightcovid19.hku.hk/content/uploads/2020/10/Image-2-390x220.jpg)
![Photo of [Cell Reports Medicine] Oral SARS-CoV-2 inoculation establishes subclinical respiratory infection with virus shedding in golden Syrian hamsters](https://fightcovid19.hku.hk/content/uploads/2020/10/fx1_lrg-e1601870075235-390x220.jpg)